What the Internet Gets Wrong About Leaky Gut
In the late 1700s, a German scientist and philosopher named Georg Christoph Lichtenberg famously wrote: “The most dangerous untruths are truths slightly distorted.” I think of this quote often in the context of the pervasiveness of a questionable “diagnosis” called Leaky Gut Syndrome in certain corners of the Internet and alternative medicine world. But before we start sorting through what’s fact and what’s fiction, we need to get grounded in some basic fundamentals of the gut’s anatomy and functions.

First, Let’s Get Acquainted With Our Gut
The intestines are essentially a long, winding tube wrapped in layers and sublayers of different types of tissue. The hollow inside of our gut where food and waste pass through is called the lumen.

The innermost layer lining the lumen is a row of cells called enterocytes; these are the cells that selectively absorb or engulf various nutrients from our food, carrying them from inside the lumen through their very own cell ‘bodies’ and out the other side, where they enter into the bloodstream, heading first to the liver and then beyond. Schematic drawing of three enterocytes. From Jutfelt F (2006) The Intestinal Epithelium of Salmonids: Transepithelial Transport, Barrier Function and Bacterial Interactions. PhD Thesis, University of Gothenburg, Sweden.
I like to picture the enterocytes like a row of Rockettes:

Standing in a row with their feet facing into the lumen of the gut, shoulder to shoulder, with their arms tightly linked to one another at the elbows. Underneath their feet is a mucus layer that forms the gut’s first line of defense. This gooey, sticky mucus helps to keep the trillions-strong population of gut bacteria inside the gut’s lumen where they belong, rather than adhering to the soles of the Rockettes’ feet—or the enterocytes themselves.
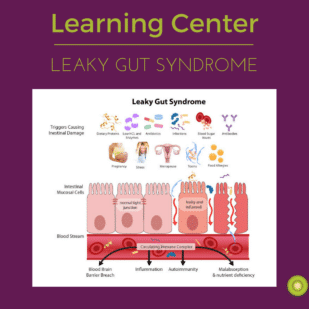
What Can Pass Between The Tight Junctions?
Nutrients that our body wants to absorb—sugars broken down from starches or fruits we’ve eaten, protein building blocks that have been liberated by our digestive enzymes from longer protein chains, fatty acids that have been liberated from fat—are selectively allowed into the body through the enterocytes themselves. But water, some electrolytes and itty bitty sugars are allowed to flow into the body as well in between the individual Rockette/enterocytes as well. The ‘linked elbows’ connecting these cells side by side (which in real life are called “tight junctions”) keep out larger molecules like larger food proteins, large sugars, bacteria or bacterial byproducts. This between-the-cells pathway is a normal way for certain types of small molecules to make it into the body.
Second Line of Defense
At the other end of the enterocytes—anchoring the Rockettes by their heads, if you will—is a layer of connective tissue. Embedded in this layer are a variety of immune cells, and they’re there to form a second line of defense to buttress the physical defenses of the mucus layer and those ‘linked-elbow-like’ tight junctions. (Evolution wasn’t about to leave it to two modest physical barriers to separate the inside of our bodies from a trillions-strong community of bacteria residing just on the other side!)
Not So Leaky After All
Should any protein fragments, bacterial toxins or even whole bacteria make it through between the cells, they encounter these immune cells, who then spring into action, causing a localized inflammatory reaction in the gut as they set about doing damage control with respect to the invaders that slipped through. Even if the gut’s immune cells failed to neutralize any stowaway proteins or compounds that slipped past its physical barriers, we have yet another line of defense: our liver. All of the blood flow from the intestines is routed first to the liver, where any bacteria, bacterial toxins or larger food proteins that don’t belong in the systemic blood circulation are filtered out and disposed of.
You May Want To Read: VITAL ORGANS: DIGESTIVE SERIES – The Liver
After all: if bacteria actually made it into the systemic bloodstream from the gut, it could create a life-threatening illness called sepsis. We humans didn’t make it this far into our evolution without backups for our backups for our backups to keep any undesirable molecules from the ‘outside world’ out of our bloodstream.
So, What Is Gut ‘Leakiness’?
For decades, scientists and clinicians have been engaged in painstaking research to understand a phenomenon called “intestinal permeability,” which is what people mean when they refer to the gut’s “leakiness.” “Permeability” is the degree to which a material or membrane allows liquids or gasses to pass through it; the opposite of permeability is ‘barrier function.’
![BallenaBlanca [CC BY-SA 4.0 (https://creativecommons.org/licenses/by-sa/4.0)]](https://www.fodmapeveryday.com/wp-content/uploads/2020/01/Leaky-Gut-Horizontal-Feature-Image-3-855x570.jpg)
As described above, in a normal, healthy gut, there is always a delicate dance between these two opposing forces: some degree of permeability is required to allow water and small molecules (like electrolytes and sugars) into the body to maintain fluid balance—but too much permeability could allow undesirable molecules described above into the sublayers of the gut, activating that inflammatory immune system reaction. When the tight junctions between our enterocytes are not functioning properly—or, in our metaphor, when those Rockettes unlink their elbows—larger particles are then able to flow in-between these cells. When foreign proteins manage to reach the sublayer beneath the enterocytes, our immune cells unleash an inflammatory torrent in response. The inflammatory state can actually worsen intestinal permeability—or leakiness—allowing more and more large molecules in through these between-the-cell pathways.
Indeed, intestinal permeability has been observed in connection with a small but growing number of conditions—the best-studied of which are Crohn’s disease and celiac disease. In the case of Crohn’s disease, the leakiness resolves when patients are given medications that stop the immune cells from releasing their pro-inflammatory substances.
‘Gut Leakiness’ Is An Observation, Not A Medical Diagnosis
Intestinal permeability is indeed a real occurrence, and the researchers who study it are still very much in the early stages of trying to understanding the complex mechanisms by which it is triggered, how best to even measure it, whether it causes certain diseases like Crohn’s—or whether it is actually being caused BY these diseases.
Studies dating as far back as the late 1980s have found that subsets of healthy, first-degree relatives of people with Crohn’s disease seem to have increased intestinal permeability—suggesting that the permeability, or ‘leakiness,’ alone is not sufficient to cause inflammation or active disease. (We do not know whether those who had increased permeability went on to develop Crohn’s disease at a higher rate that those who did not, as it has not yet been studied.)
Animal studies in which intestinal permeability was induced have similarly found that the permeability alone seems not to produce disease symptoms or cause any other type of disease, either. In other words, leakiness alone is not pathological.
Leaky Gut Syndrome Is A Marketing Term, Not A Medical Term

Given how little is known about the complex phenomenon of intestinal permeability—its causes and its significance to human health– I can only imagin how annoyed the scientists who study it must be when confronted with the heavily marketed, pseudoscientific diagnosis of “Leaky Gut Syndrome.” In marketing “Leaky Gut Syndrome,” opportunistic practitioners have seized on a grain of truth from other peoples’ research, slapped a catchy label onto this barely understood observation, and spun a frightening and incredibly over-simplistic story about all the symptoms that this leakiness can cause.
For example, in a typical online description of so-called Leaky Gut Syndrome, intact food particles and whole bacteria are said to be seeping out of our inflamed intestines, floating around in our bloodstream, and causing everything from headaches, fatigue, ‘brain fog’ and joint pain to food sensitivities, depression, diarrhea, bloating and constipation.
Why Do You Think You Have Leaky Gut?
Typically, practitioners who dispense diagnoses of Leaky Gut Syndrome are also likely to market various regimens that purport to cure it. There is no shortage of protocols being touted online to this end, often involving some combination of elimination diets, bone broth, probiotics and extensive supplement regimens.
You May Want To Read: Don’t Fall For These 5 IBS Diet Fads!
As for what remedies are supported by actual scientific evidence, the only known way to reduce intestinal permeability is to identify and treat the underlying cause of it. In the case of Crohn’s disease, that often means medications which stop immune cells from releasing inflammatory compounds (though currently, a variety of diets are being investigated to determine whether diet modification has the potential to induce remission as well as medications in certain patient groups.) In celiac disease, it means removing gluten from the diet. As for other potential causes, the jury is still out.
There is no evidence to support any benefit of commonly touted supplements like L-glutamine powders, ‘herbal microbials,’ digestive enzymes, or probiotics for reducing intestinal permeability. If I were a researcher who had devoted my professional career to studying intestinal permeability, I’d be pretty angry at those who commandeered my research to enrich themselves on supplement sales.
Save Your Money

And if I were a patient with actual health problems—whether chronic diarrhea, bloating, fatigue or headaches—I might be pretty angry, too, that someone charged me hundreds of dollars on baseless tests and exorbitant supplement regimens—to cure a ‘syndrome’ that no one has even proved actually exists. Prospective patients often ask me whether I “believe in” Leaky Gut Syndrome. It’s a strange question to ask an evidence-based healthcare professional. In science, belief doesn’t really come into play. We speak about data, evidence and degrees of certainty. Some things have strong evidence to support them, some have moderate evidence to support them, some have weak evidence to support them and others have no evidence to support them.
The evidence to support the existence of intestinal permeability appears pretty strong. The evidence to support a medical condition called “Leaky Gut Syndrome” that causes migraines, mood disorders, brain fog, digestive disturbances and a laundry list of other symptoms? There is none.
In my experience, people who suffer from symptoms that science cannot yet explain satisfactorily are especially vulnerable to those who swoop in assuredly with the simple, catch-all explanation for all that ails—a Leaky Gut Syndrome “diagnosis.”
We Understand The Desire For A Diagnosis
To be offered a clear, definitive treatment protocol after a prolonged search for answers can feel like a prayer has been answered. I do understand why many people choose to believe, even in the absence of evidence.
Save this article
Tests For Leaky Gut
“What about that test I was given that said I had “Leaky Gut”?”
There are a variety of tests which claim to ‘diagnose’ leaky gut being marketed by labs that cater to naturopaths, chiropractors and integrative/functional medicine practitioners—from a urine test that measures the ratio of sugars called lactulose and mannitol to blood or stool tests that measure a protein called zonulin. (Most lab results will include an asterisk and fine-print disclaimer that these tests are not FDA-approved to diagnose any disease.) The premise behind all of these tests is to look for evidence that the body has absorbed some larger molecules that it shouldn’t have been able to absorb if the gut’s barrier function were working properly.
The problem is, however, that as of the time of this writing, there is no scientifically validated clinical test to measure intestinal permeability in the general population.
Notice my deliberate use of the term “measure” rather than “diagnose.” That’s because, if you’ll recall, that *some* degree of intestinal permeability is normal, so researchers who study this stuff are simply looking to understand the degree of permeability a person may have, and any changes in response to various interventions.
To be sure, there are many different methods that researchers employ in their work toward quantifying the degree of a gut’s permeability. A 2016 paper published in the European journal Neurogastroenterology & Motility reviewed a long list of them, explaining the advantages and drawbacks of a variety of different approaches that are currently used. The authors described various measurement approaches and explained their respective limitations and the potential for misinterpretation inherent to each. Of note, no stool tests were mentioned in the list of current methods being used to measure intestinal permeability in the research community.
They concluded that researchers would be well advised to use a few different methods and compare their results, since there is no single, extremely reliable measurement method.
Is Intestinal Permeability Even A Problem?
“Should intestinal permeability even be treated?”
Even if there were a validated way for your doctor to accurately and reliably measure your degree of intestinal permeability—as there may well be in the not-too-distant-future– the question remains: what should one do with the results?
In the absence of an obvious underlying disease, it’s not yet clear that increased intestinal permeability even signals something is amiss that must be acted upon.
So far, animal studies suggest that increased intestinal permeability alone is not enough to induce disease, and the studies of healthy relatives of people with Crohn’s disease are walking around oblivious to the fact that their intestines are more permeable than other peoples’ certainly raises the possibility that a whole lot of healthy people could be similarly walking around with increased intestinal permeability and nothing may ever come of it.
For now, the point is somewhat moot, as there’s no known way to reduce intestinal permeability other than correcting its underlying cause. The one and only trial of a medication designed to correct the faulty tight junctions in people with celiac disease (larazotide) failed to demonstrate a benefit. Researchers are currently investigating other therapies that would target some of the proteins which regulate tight junctions—but nothing is yet known.
Dr. Google Says Leaky Gut Is a Huge Issue

The internet-fueled obsession with ‘healing leaky gut’ reminds me that sometimes our attempts to ‘fix’ something that we don’t understand can backfire. Here’s a case in point: Not long ago, it was common practice for athletes and weight lifters to take antioxidant supplements following their workouts. The rationale seemed logical: scientists had long observed that exercise resulted in the development of “free radicals”—or, unstable, negatively charged molecules that can damage cells. Since antioxidants neutralize free radicals, it seemed common-sensical for athletes to take antioxidant supplements like Vitamins C and E to help protect their cells from the risk of oxidative damage induced by exercise. That is, until researchers finally figured out that those supposedly harmful free radicals had a beneficial purpose: they stimulated various physiological processes that increased endurance and enhanced post-exercise muscle repair.

A series of randomized, controlled studies conducted in both endurance athletes and weight lifters revealed that taking antioxidant supplements in an attempt to ‘fix’ something that appeared to be damaging actually resulted in worse outcomes compared to leaving our body’s evolution-driven-devices alone—and not taking the supplements. In this case, the worse outcome was reduced strength building and endurance in antioxidant-supplementing athletes compared with those who did not supplement.
Bone Broth, Cleanses, Collagen, Nightshades, Lectins & Beyond
Elimination diets more likely to deplete the gut’s barrier function, not improve it.
An ironic twist in the story of how ‘gut leakiness’ was spun into ‘Leaky Gut Syndrome’ is the possibility that a commonly marketed remedy for the latter is a type of severely restricted elimination diet that may actually worsen gut barrier function.
There are many versions of ‘gut healing’ diets being peddled online that claim to reduce intestinal inflammation or heal a “leaky gut”—none of which have any scientific basis.
Some regimens call for multi-day ‘cleanses’ consisting only of bone broth, under the rationale that fasting provides “gut rest” that helps heal inflammation, while the collagen in bone broth should somehow repair porous gaps between gut cells. Fact check: the tight junctions are regulated by a variety of different proteins in a very complex manner; they’re not held together by bands of collagen that need to be patched up. Other regimens call for elimination of a variety of plant-based foods that their advocates deem to be inflammatory: from grains and legumes to “nightshade” vegetables, foods high in lectins or higher sugar fruits.
You Might Be Eliminating Healthful Foods
Many such ‘gut healing’ protocols therefore result in a diminished intake of total fiber—or at least diminished dietary variety of fermentable, prebiotic and/or soluble fibers that are especially good at nourishing the gut microbiota. Yes—it’s unfortunately true that eating fewer FODMAP-containing foods adversely impact the diversity of the gut microbiota… which is why followers of the diet are encouraged not to get stuck in the strictest elimination phase indefinitely, and why consuming even those very small, tolerable portions of high FODMAP foods is a wise strategy for people following the diet longer term!
You May Want To Read:
- The Low FODMAP Diet Elimination Phase: Short & Sweet for Your Health!
- The “Low FODMAP Diet” is NOT Just About The Elimination Phase
But here’s the rub: research has shown that diets devoid of fiber actually deplete the mucus barrier—our gut’s first physical line of defense. This is because our gut bacteria depend on a steady and diverse stream of dietary fiber as their food source. When we starve the gut bacteria, they begin degrading the mucus lining as an alternate source of nourishment. Why is this a problem? Because a depleted mucus layer lining the gut makes a person more susceptible to infection by disease-causing bacteria. In other words, if you’re truly worried about your gut’s leakiness, you may be better off looking to expand the variety of whole, plant-based foods you eat—not culling it.
Just how diverse a diet is optimally nourishing for gut health?
Research from the American Gut project at University of California San Diego’s School of Medicine suggests that the most diverse gut microbiomes in their data set of over 17,000 stool samples belonged to people who reported eating thirty or more different plant species per week. Relatedly, the extensive review of over 1,900 research studies that was used to create a Dietary Inflammatory Index (DII)—which assigns evidence-based scores to dozens of foods and nutrients to quantify how pro-or-anti inflammatory they are—ranks fiber as among the most anti-inflammatory nutrients we could consume.
You May Want To Read: TOP 5 HIGH FIBER LOW FODMAP FOODS
All of this is to say: for good gut health, the scientific evidence suggests that eating the most diverse array of plant-based foods that you can comfortably tolerate is a pretty sound strategy. Adopting a severe elimination diet in the service of good gut health may be inadvertently shooting yourself in the proverbial foot.
You May Want To Read: Intro To Leaky Gut
The Takeaway
The human species has made it quite far with its elegant evolutionary adaptations, and there is a large, global community of brilliant scientific thinkers who have devoted their lives to understanding the intricacies of how our bodies function… and sometimes malfunction.
The work of scientific inquiry is deliberate, painstaking, and—in our instant gratification culture—sometimes frustratingly slow.
It can be tempting to take a bit of scientific fact and run with it—extrapolating conclusions that haven’t yet been demonstrated, using what seems to be ‘common sense’ to generate solutions to perceived problems, mistaking an interesting hypothesis for bona fide fact.
From where I sit, it seems that most of what is marketed as Leaky Gut Syndrome corresponds to one or more of these fallacies.
Read All Of Tamara’s Articles:
- What Conventional Wisdom Gets Wrong About Bloating
- Timing of Digestive Symptoms: What It Means
- What Is Leaky Gut Syndrome?
- Everything You Want to Know About Farting*
- 5 Reasons to Skip Gut Microbiome Testing – For Now
- Exclusive Interview with Dietitian Tamara Duker Freuman
- Q & A With The Author Of “Regular, The Ultimate Guide to Taming Unruly Bowels and Achieving Inner Peace”








You could do a powerpoint webinar for dietitians on this. Nice job!
Thank you so much for the note – and we will make sure Tamara knows! We felt this was a very important topic to cover and we knew that she would write an article the way it needed to be presented.
Superb and very helpful. Tamara’s writings are so helpful. Her book is excellent.
Eileen, we couldn’t agree more! Thank you for letting us now that you found this article insightful. Please feel free to share far and wide – it needs to be read!
This is a great article! Since the author points out the issue with current tests: “there are a variety of tests which claim to ‘diagnose’ leaky gut being marketed by labs that cater to naturopaths, chiropractors and integrative/functional medicine practitioners—from a urine test that measures the ratio of sugars called lactulose and mannitol to blood or stool tests that measure a protein called zonulin.
Maybe it’s appropriate to address this post on your site that makes it sound like the testing is appropriate and accurate? https://www.fodmapeveryday.com/intro-leaky-gut/
“There are several ways to test for leaky gut. One of the most common methods is the lactulose/mannitol urine test. This is a simple, non-invasive test that measures the amount of specific sugars in your urine. Using this information, a ratio is calculated that can give you an idea of whether leaky gut is present or not.”
Thank you for being so thorough! These articles were written by two different RDs with different points of view and approaches to their patient care. It is always up to the patient to do their due diligence and make decisions about their own medical care. Also please note that the quote says “give you an idea”, which is not an absolute, and was deliberate wording.
Curious about gluten and dairy and their supposed link to Hashimotos hypothyroiditis. I still struggle to understand this link. Why do so many people insist gluten/dairy free is necessary for this thyroid condition? Thank you for this great article that non medically trained people can understand;)
Hib Jessica, we have an answer for you from Tamara: “You can respond from me as follows:
According to my read of the scientific literature, the reason it is speculated that a gluten free diet may benefit people with autoimmune thyroiditis is because Hashimoto’s and celiac disease often ‘cluster,’ meaning that people who have one are genetically predisposed to also have the other. It is speculated that undiagnosed/untreated celiac disease may unleash an autoimmune inflammatory process that can also trigger onset of (or worsen per-existing) autoimmune thyroiditis, and it is common practice to screen for celiac disease in patients newly diagnosed with thyroiditis. In the absence of celiac disease or at least elevated anti-gliadian antibodies, it is not clear that there are much data to support an independent benefit of a gluten free diet for people with Hashimoto’s. Research is quite lacking, though.
There’s not much of anything in the scientific literature exploring a direct link between dairy and Hashimoto’s, though if I had to guess at the basis of this common recommendation, I’d say the likely connection is that diets very high in iodine (or perhaps more accurately, bodies that fail to adequately excrete dietary iodine efficiently) have been linked to increased frequency of thyroiditis. Since dairy has historically been a significant contributor of iodine to the American diet, perhaps this is the connection? (Though its worth noting that the iodine content of dairy has dropped significantly since the 1970s with changes to animal feed formulations common in the dairy industry). It’s an interesting question, though, because I don’t see a lot of talk about limiting other high iodine foods like seafood, seaweed and iodized salt… why is dairy still singled out? if the dairy/iodine connection is indeed a factor in Hashimoto’s, though, it would have nothing to do with intestinal permeability but seems more likely related to wonky kidney function, since ~90% of ingested iodine is excreted in the urine.”
Thank you for posting and promoting this article. It is refreshing to read something based on scientific evidence that calls out the marketing hype. This article also contains the best explanation I’ve seen on the benefits of dietary fibre.
Sharon, thank you so much for taking the time to write. We love our RD team and are so grateful for their hard, evidence based work – and we are so glad you have found our community. Enjoy looking through the other articles – there is a lot to read!